Production Operators work in a (non-chemical or non-biochemical) manufacturing environment to transform individual parts, sub-assemblies, and materials into distinct products (i.e. cars, phones, TVs, computers, cans, and machine tools) often on an assembly line. They oversee machinery, handle equipment setup, maintenance, and adjustments, monitor product standards, and enforce safety protocols.
How GetReskilled’s Online Learning Compares to Zoom Lectures, Self-Paced Courses, and Short Seminars
Production Operators work in a (non-chemical or non-biochemical) manufacturing environment to transform individual parts, sub-assemblies, and materials into distinct products (i.e. cars, phones, TVs, computers, cans, and machine tools) often on an assembly line. They oversee machinery, handle equipment setup, maintenance, and adjustments, monitor product standards, and enforce safety protocols.
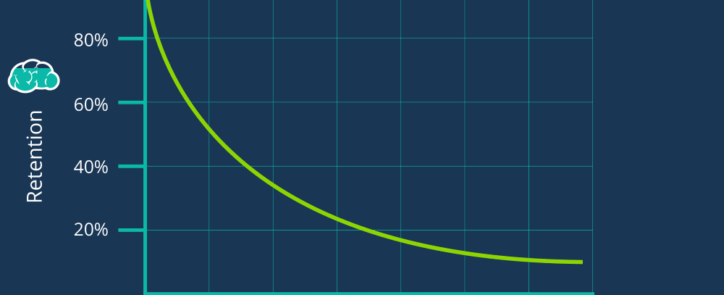
The Forgetting Curve in Pharma Manufacturing
Production Operators work in a (non-chemical or non-biochemical) manufacturing environment to transform individual parts, sub-assemblies, and materials into distinct products (i.e. cars, phones, TVs, computers, cans, and machine tools) often on an assembly line. They oversee machinery, handle equipment setup, maintenance, and adjustments, monitor product standards, and enforce safety protocols.
What is a Production Operator and How Can You Become One?
Production Operators work in a (non-chemical or non-biochemical) manufacturing environment to transform individual parts, sub-assemblies, and materials into distinct products (i.e. cars, phones, TVs, computers, cans, and machine tools) often on an assembly line. They oversee machinery, handle equipment setup, maintenance, and adjustments, monitor product standards, and enforce safety protocols.
What is a Process Operator and How Can You Become One?
Process Operators operate and monitor chemical or biochemical manufacturing processes, especially continuous flow ones on an industrial scale to turn raw materials such as milk, oil or natural gas (using heat, cold, pressure, or a chemical agent) into an end product (e.g. butter, beer, milk formula, drugs, vaccines, paint, etc.). These individuals play a crucial role in ensuring the smooth and efficient running of manufacturing or production processes.
How long does it take to get a job in the pharma industry?
How long does it take to go from making a job application to starting your first day at work in a pharmaceutical or medical device company? Of course, it varies from company to company and depends on the type of job you are applying for but the answer is commonly “much longer than expected”! Getting a job in this sector takes time and requires a significant amount of effort, preparation and waiting. The recruitment processes are generally multistep and rigorous with multiple checks and balances.
What Types of Jobs and Careers are there in the Pharmaceutical and Medical Device Industry
By: Donagh Fitzgerald and Claire Wilson. Last Updated: July 2024 Image: Cambridge Clinical Laboratories There are a huge variety of job roles available from laboratory-based research and development, clinical trials, regulatory affairs, drug safety, manufacturing, engineering, quality assurance, quality control, and validation all the way [...]
What is a Manufacturing Technician and what do they do?
By: Donagh Fitzgerald BEng and Claire Wilson BSc. Last Updated: July 2024 Manufacturing Technicians set up, run and monitor (non-chemical or non-biochemical) manufacturing processes to transform individual parts, sub-assemblies, and materials using machining, cutting, assembling, 3D printing, bolting, welding, forging, etc into distinct products (cars, phones, TVs, [...]
What is an Instrumentation Engineer? What Do They do?
By: Donagh Fitzgerald BEng and Claire Wilson BSc.. Last Updated: July 2024 Instrumentation Engineers are responsible for planning, installing, monitoring and maintaining control systems and machinery within manufacturing environments. They typically work with control processes that use sensors to provide feedback. As a result, that can also [...]
What is a Biochemist and what to they do?
By: Donagh Fitzgerald B.Prod Eng and Claire Wilson BSc. Last Updated: July 2024 Biochemists are typically lab-based professionals who study and explore the chemical processes occurring within living things. They are employed across a wide range of industries and within academia. Many Biochemists are employed within research but [...]
What is a Microbiologist and what do they do?
By: Claire Wilson. Last Updated: April 2024 What is a Microbiologist? A Microbiologist is a scientific professional who studies microorganisms. They play a key role in pharmaceutical or medical device manufacturing by testing to monitor levels of microbial contamination at all stages of the manufacturing process. [...]
What is a Facilities Engineer and what do they do?
By: Donagh Fitzgerald B.Prod Eng and Claire Wilson BSc. Last Updated: July 2024 Image: GSK Facilities Engineers work in a number of indoor environments including manufacturing facilities, power stations, hospitals, and offices. They can have an extremely broad remit covering the plant maintenance or infrastructure of the [...]
Documentation Specialist: Role, Responsibilities & Career Path
By: Donagh Fitzgerald B.Prod Eng and Claire Wilson BSc. Last Updated: July 2024 What is a Documentation Specialist? A Documentation Specialist is responsible for managing, maintaining, and optimizing a company's documentation processes, ensuring compliance with industry regulations. In highly regulated industries like pharmaceuticals, biotechnology, and medical devices, [...]
What is a Manufacturing Engineer and How Can I Become One?
By: Donagh Fitzgerald BEng and Claire Wilson BSc. Last Updated: July 2024 Photo: Sandra Goettisheim, KIT Manufacturing Engineers design, implement and optimise (non-chemical or non-biochemical) manufacturing systems, particularly machining and assembly processes to turn individual parts, sub-assemblies, and raw materials into finished distinct products (cars, phones, [...]
What is a Calibration Technician and what do they do?
By: Donagh Fitzgerald B.Prod Eng and Claire Wilson BSc. Last Updated: July 2024 A Calibration Technician is responsible for the calibration, routine inspection, testing, maintenance and repair of instruments, meters, gauges and other testing and measuring equipment. The goal of a Calibration Technician is to ensure the accuracy [...]
What is a Production Supervisor and How Can I Become One?
By: Donagh Fitzgerald and Claire Wilson. Last Updated: July 2024 Production Supervisors are directly responsible for overseeing and organizing the equipment, staff, and processes on a production floor. They will oversee scheduling and routine production activities as well as acting as the first line of support for [...]
What is a Process Engineer and How Can You Become One?
By: Donagh Fitzgerald B.Prod Eng and Claire Wilson BSc. Last Updated: July 2024 Process Engineers design, implement and optimise chemical and biochemical processes, especially continuous flow ones on an industrial scale to turn raw materials such as oil, natural gas or milk (using heat, pressure or a [...]
What is a Process Technician and How Can You Become One?
Process Technicians operate and monitor chemical or biochemical manufacturing processes, especially continuous flow ones on an industrial scale to turn raw materials such as milk, oil or natural gas (using heat, cold, pressure, or a chemical agent) into an end product (e.g. butter, beer, milk formula, drugs, vaccines, paint, etc.). Process technicians typically have some training in chemical engineering or chemistry.
What is a Project Engineer and How Can You Become One?
By: Donagh Fitzgerald B.Prod Eng and Claire Wilson BSc. Last Updated: July 2024 A Project Engineer manages technical or engineering projects. They work with stakeholders at all levels, with direct responsibility for budgeting, personnel and project planning and making sure the project is finished on time and on [...]
What is a Quality Assurance Specialist and How Do You Become One?
By: Donagh Fitzgerald B.Prod Eng and Claire Wilson BSc. Last Updated: March 2025 Image: Solar Career Map Quality Assurance Specialists check the implementation of the quality system and conduct quality assurance audits. They create and implement well-defined standards, procedures and checklists to reduce variation and ensure [...]
What is a Quality Control Analyst and How Do You Become One?
By: Donagh Fitzgerald B.Prod Eng and Claire Wilson BSc. Last Updated: July 2024 Quality Control Analysts check or test the product of a manufacturing process to make sure that it meets predefined quality or safety standards. The completion of this quality check ensures that the final product is [...]
What is a Packaging Operator and How Can You Become One?
By: Donagh Fitzgerald and Claire Wilson. Last Updated: July 2024 A Packaging Operator is responsible for taking the finished product of a manufacturing process and ensuring that it is packaged in line with company and industry standards, making it ready for sale or distribution. Some Companies [...]
What is a Maintenance Technician and How Can You Become One?
By: Donagh Fitzgerald B.Prod Eng and Claire Wilson BSc. Last Updated: July 2024 A Maintenance Technician performs routine maintenance of equipment, machinery and manufacturing facilities and helps troubleshoot and repair any mechanical or electrical problems when they arise. According to the BLS, the median annual wage for [...]
What is a Quality Engineer and How Can You Become One?
By: Donagh Fitzgerald B.Prod Eng and Claire Wilson BSc. Last Updated: April 2024 Image: Ingersoll Rand Quality Engineers work to ensure the overall quality of manufactured products. They test processes, monitor quality standards, create documentation, devise quality tests and define the criteria a test result should [...]
What is a Laboratory Technician and what do they do?
By: Donagh Fitzgerald B.Prod Eng and Claire Wilson BSc. Last Updated: July 2024 Laboratory Technicians assist scientists with research, testing, and conducting experiments. Their tasks can vary greatly between roles but are almost always laboratory-based. They may work alone on specific tasks but are usually part of a [...]
What is an Automation Engineer and How Can You Become One?
By: Donagh Fitzgerald B.Prod Eng and Claire Wilson BSc. Last Updated: July 2024 Image: Festo An Automation Engineer uses technology to improve, streamline and automate manufacturing, electricity generation, warehouse distribution, mining and many other processes to reduce the need for human intervention and maximise efficiency. They are [...]