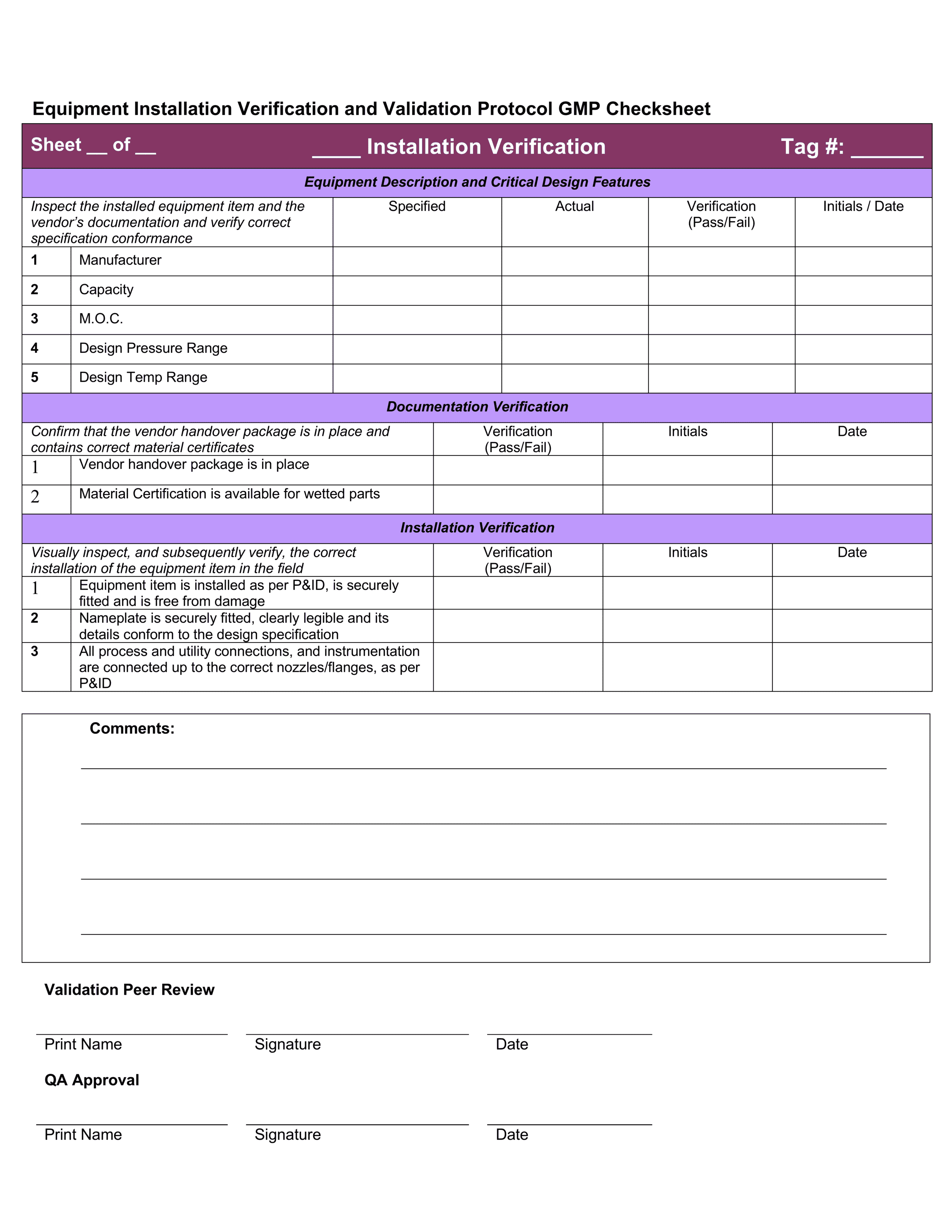
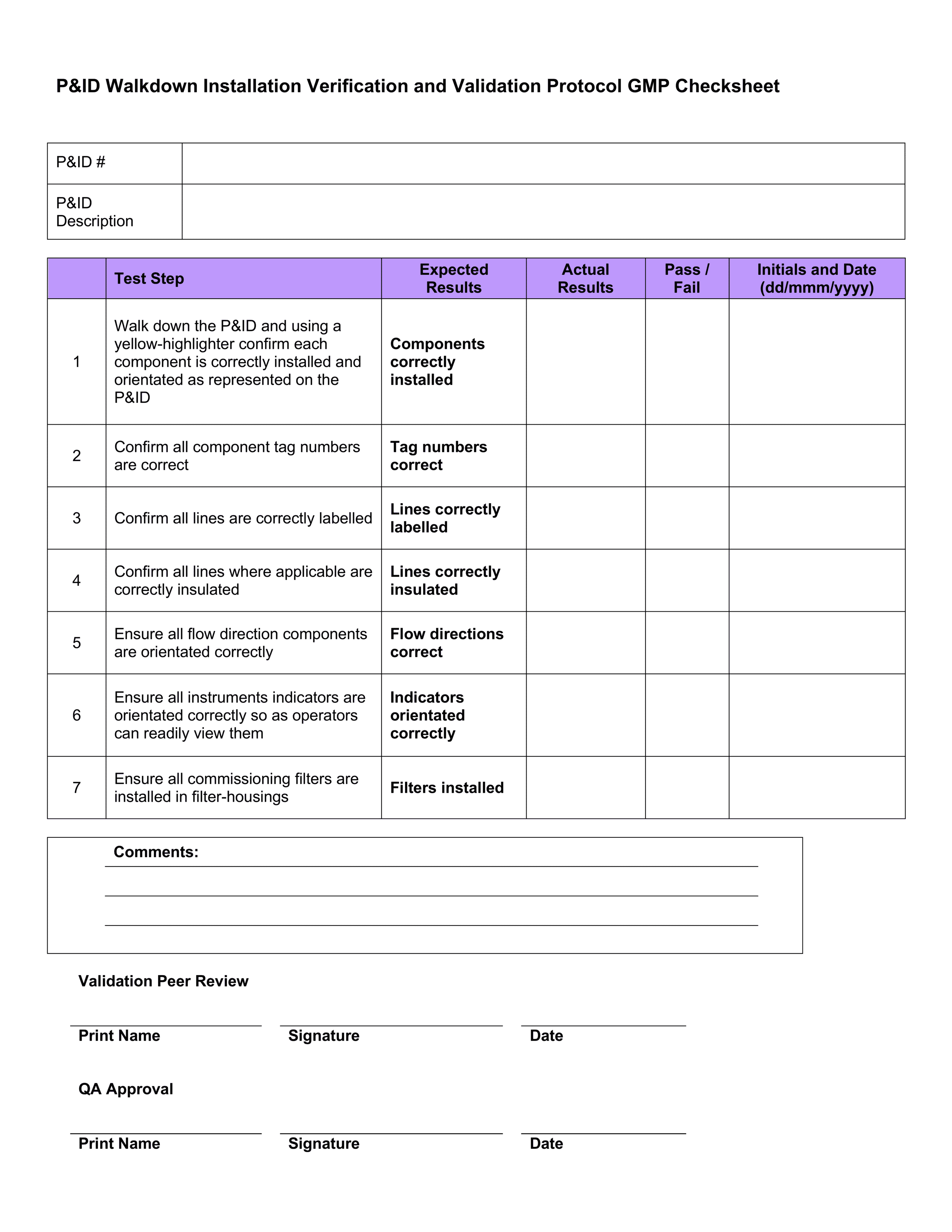
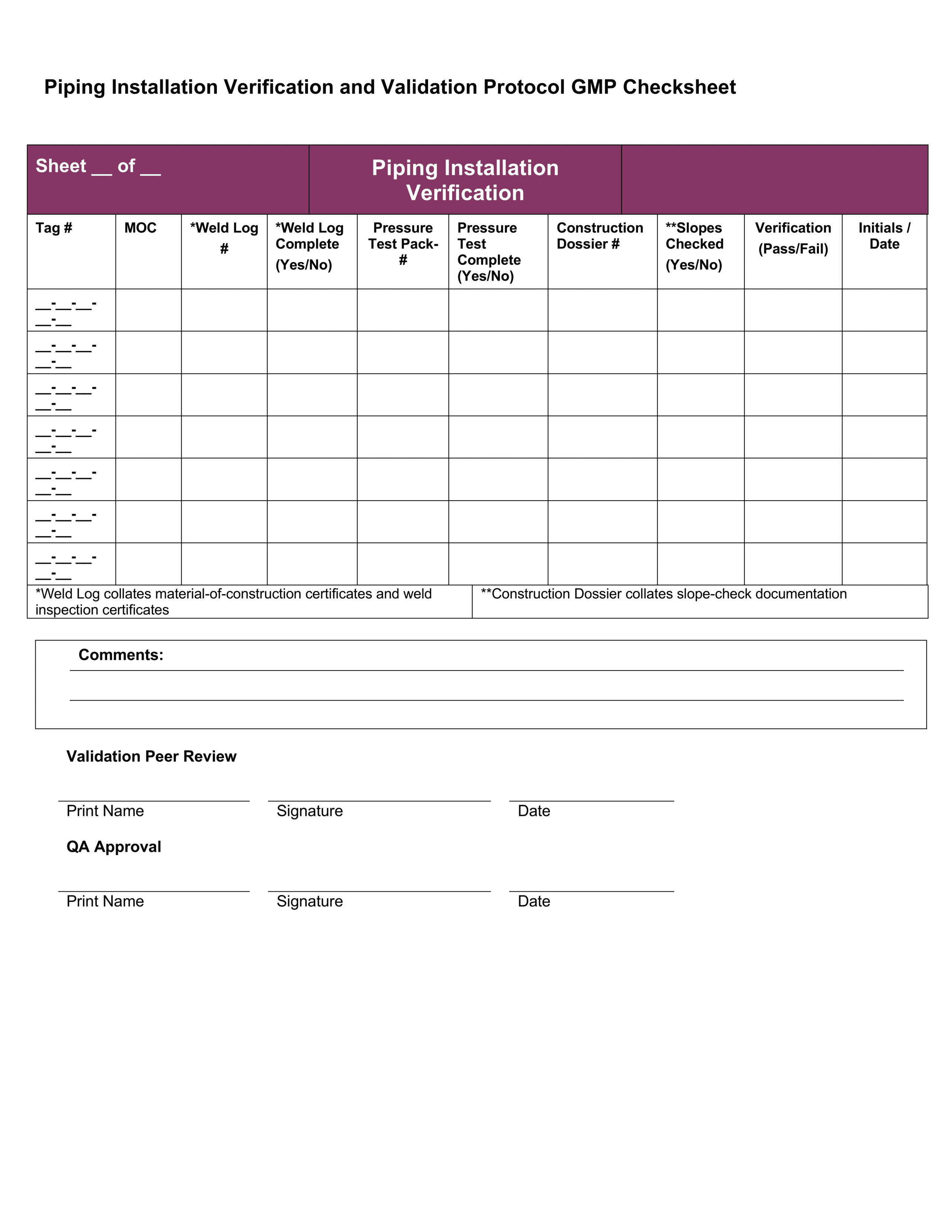
Download 4 Professional IQ OQ PQ Templates
You can use them right now to help with your qualification and validation projects. Save them on your hard drive and follow the detailed instructions to easily customize them.
P&ID Walkdown Installation Verification and Validation Protocol GMP Checksheet
Instrumentation Installation Verification and Validation Protocol GMP Checksheet
These testing templates are collated in a document called an Equipment Validation Protocol which is a written plan stating how validation will be conducted. It details factors like:
- Test scripts and methods – telling you the steps involved in conducting a test
- Test parameters and acceptance criteria – defining acceptable test results
- Test checksheets – documenting and recording the test results
- Product characteristics – showing what your system is looking to achieve/produce
- Production equipment – detailing the equipment necessary
- Final approval – documenting that the validation process has been successfully carried out
Equipment Validation Training Course
Not sure how to perform an IQ IQ PQ or to populate an equipment validation protocol? Then check out our 15-week Equipment Validation Training Course (ONLINE) – which breaks the process step by step and will retrain you for entry-level validation, CQV and C&Q roles.