At a Glance
- Over 10,700 people directly employed in Pharmaceutical and Medical Device Manufacturing
- Home to manufacturing sites for four of the world’s top 15 pharmaceutical companies
- More than 33 companies have manufacturing sites
- Many more have R&D, corporate and business support functions
Dublin consistently ranks as the number one county for available Pharmaceutical and Medical Device jobs in Ireland.
Read on to find out more…
BTW, if you are interested in a career in the Pharmaceutical or Medtech industries or are considering upskilling for a better job or a promotion, check out our range of pharmaceutical courses.
List of 33 Pharmaceutical and Medical Device Manufacturing Companies in Dublin
| Company | Product | Employees |
|---|---|---|
| AbbVie | Pharma - Manufacturing | 400 |
| Astra Zeneca (Alexion) | Pharma - Manufacturing | 500 |
| Amgen | BioPharma - Biologics Manufacturing | 670 |
| APC | Pharma - Vaccine Manufacturing | 260 |
| Astellas | Pharma - API Manufacturing | 70 |
| Baxter | Pharma - Compounding Facility | 200 |
| B. Braun | Medical Devices - Wound Care | 50 |
| Bimeda Ireland | Animal Health | 80 |
| Bristol Myers Squibb | BioPharma - Biologics Manufacturing | 650 |
| Connexicon Medical | Medical Devices - Wound Closure | 15 |
| embecta (Formerly BD) | Medical Device Manufacturing | 350 |
| Grifols | Pharma - Manufacturing | 300 |
| Guerbet | Pharma - API Manufacturing | 130 |
| Helsinn | Pharma - Manufacturing | 180 |
| Ipsen | Pharma - API Manufacturing | 165 |
| Jabil | Medical Devices - Manufacturing | 300 |
| Keenova (Formerly Mallinckrodt | Medical Devices - Engineering | 120 |
| Kypha | Diagnostics - Healthcare | 10 |
| LEO Pharma | BioPharma - Biologics Manufacturing | 650 |
| Meditec Medical | Medical Devices | 10 |
| MSD | BioPharma - Biologics Manufacturing | 400 |
| Niche Generics | Pharma - Manufacturing | 80 |
| Pfizer | BioPharma - Biologics Manufacturing | 2400 |
| Siemens Healthineers | Medical Devices - Medical Imaging | 450 |
| SK Biotek | Pharma - API Manufacturing | 400 |
| Takeda | BioPharma - Stem Cell Therapy | 200 |
| Viatris | Dry Powder Inhalers | 300 |
| Viatris - Damastown | Pharma - Manufacturing Oral Solid Doses | 320 |
| West | Healthcare, Med Devices and Assemblies | 650 |
| Xeolas Pharmaceuticals | Pharma - Manufacturing | 65 |
| Zoetis | Animal Health | 75 |
| Total | 10536 |
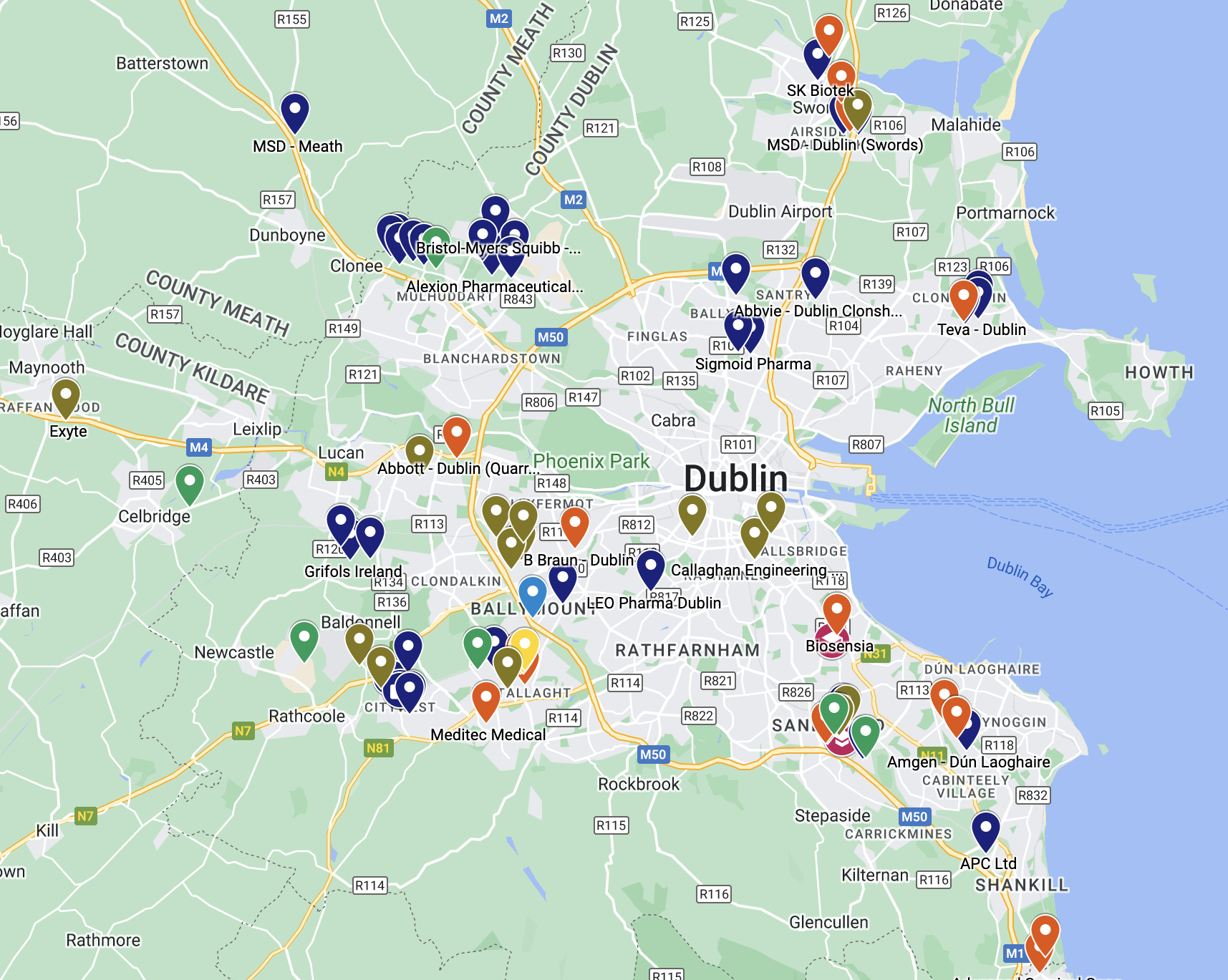
View Pharma Companies on a Google Map
Find all pharmaceutical and medical device manufacturing facilities in Dublin using this interactive Google Map.
Many of these companies also have offices in County Dublin that support business operations, commercial activities, supply chain management, and more.
Non-Manufacturing sites
Many other companies have significant non-manufacturing operations in areas such as R&D, corporate and business support, clinical research, clinical trials, etc.
🔬 Research & Development
- APC & VLE Therapeutics – Research
- Carrick Therapeutics – Research and Development
- Gilead Sciences – Global Pediatric Drug Development
🏢 Corporate Headquarters & Commercial Offices
- AbbVie – Citywest – Commercial Offices
- Abbott – Support Operations
- Aspen Pharmacare – Pharmaceutical Operations
- Bayer – Commercial
- Boehringer Ingelheim – Commercial
- Cardinal Health – Commercial
- Horizon Therapeutics – Headquarters
- Jazz Pharmaceuticals – Headquarters
- Johnson & Johnson – Support Operations
- Kyowa Kirin – Commercial
- Medtronic – Dublin – Corporate Headquarters
- MSD – Commercial Head Office
- Novartis – Global Services
- Pfizer – Citywest – Customer Contact Centre
- Pfizer – Ringsend
- Regeneron – Business Operations
- Roche – Commercial Operations
- Sanofi – Commercial
- Takeda – Citywest Business Campus
- Takeda – Baggot Street – Global Shared Services
- Uniphar Group – Healthcare Services
🧪 Clinical Research & Trials
- Icon – Clinical Trials Management
- Parexel International – Clinical Research
Pharma & Medical Device Job Announcements in Dublin
There has been much investment and a number of job announcements in Dublin recently. Some of the most significant include:
- Pharma company Grifols to create 200 new jobs at its Dublin site
- Pfizer to invest over $1.26 billion in 500 Irish jobs boost
Here is a look at some of the major companies.
Top Pharmaceutical Companies in Dublin
Alexion/AstraZeneca
- Activities: Pharma – Manufacturing
- Employees in Dublin: 500
Alexion has operated a site in Blanchardstown, Dublin since 2013. This has previously housed the Global Supply Chain and Operations Headquarters, as well as a packaging and warehousing plant.
In 2015, the company announced a €450m investment to build a new large-scale biologics manufacturing facility on the site. This was the company’s first biologics manufacturing site outside of the US. At the time, this was the single biggest investment a pharmaceutical company had made in Ireland.
In 2020, Alexion’s College Park site in Dublin was named Pharma Industry Company of the Year.
In 2021, Alexion was acquired by the British pharmaceutical company, AstraZeneca. Since then, two investments (of $360m and $68.7m) have been announced for the Dublin site.
Bristol Myers Squibb
- Activities: BioPharma – Biologics Manufacturing
- Employees in Dublin: 650
Bristol Myers Squibb (BMS) has had a presence in Ireland for the last 50 years. In Dublin, the company now operates a Biologics Manufacturing site (in Cruiserath) and an External Manufacturing Division.
Construction of the Cruiserath Biologics Manufacturing site began after a €720m investment in 2015. Operations started in 2018 and the site now employs 400 people. This is the company’s first biologics manufacturing facility outside of the US. The facility is highly flexible – it is initially manufacturing a single product at a time, moving to 3-5 campaigns per year.
The Cruiserath operation was ranked 14th in the Great Places to Work category of “Best Large Workplaces in Ireland 2023”.
In early 2024, BMS was granted planning permission for a new manufacturing facility on the Dublin site. This is expected to create 300 new jobs once complete.
Take a look inside Bristol Myers Squibb’s Dublin manufacturing site…
Leo Pharma
- Activities: BioPharma – Biologics Manufacturing
- Employees in Dublin: 650
Leo’s Dublin manufacturing site was established in 1959. The Finished Goods Manufacturing site manufactures dermatology products for global distribution. The factory operates 24 hours a day, 5 days per week (“24/5”) and produces approximately 72 million units per year. Products manufactured here include topical creams, gels, ointments, and gauze.
There are also commercial activities based in the Dublin site.
Pfizer
- Activities: BioPharma – Biologics Manufacturing
- Employees in Dublin: 2400 in manufacturing
Pfizer is the largest pharmaceutical company in the world (based on 2022 revenue). It was also one of the first pharmaceutical companies to invest in Ireland, in 1969.
Pfizer has a range of commercial and business sites in Dublin, but it also has the Grange Castle manufacturing site.
Pfizer’s Grange Castle site is one of the company’s leading biotechnology manufacturing sites and represents the company’s single biggest investment in Ireland (€1.8bn). Opened in 2005, the facilities cover more than 1 million square feet, making it “one of the largest integrated biopharmaceutical plants in the world”.
Grange Castle is responsible for producing a wide range of biologics and vaccines (including finished vaccine products in pre-filled syringes and the company’s Covid-19 vaccine). The site also manufactures products for clinical trials.
The Quality Control laboratories in Grange Castle provide support to other Pfizer manufacturing sites around the world. There are significant research and development activities in laboratories here as well.
In December 2022, Pfizer announced a huge $1.2bn investment in their Grange Castle site. Once built, the new facility will double the biologic drug substance manufacturing capabilities. This is the company’s largest single investment in Ireland to date and is expected to bring 400-500 permanent new jobs once completed.
SK Biotek
- Activities: Pharma – API Manufacturing
- Employees in Dublin: 400
In January 2018, SK Biotek bought the Active Pharmaceutical Ingredient manufacturing facility in Swords formerly owned by BMS. In doing so, it became the first South Korean pharmaceutical company to invest in Ireland. The site’s 360 staff become SK Biotek employees.
The company has plans to develop a stand-alone Contract Development Manufacturing Organisation.
In June 2022, the company announced $35m investment for the first phase of expansion of the Dublin site.
Take a look inside SK Biotek’s Swords manufacturing facility…
Top Medical Device Companies in Dublin
embecta (Formerly BD Medical)
- Activities: Medical Devices – Pen Needles
- Employees in Dublin: 350
embecta has operations in Dun Laoghaire, Dublin
Having operated as a syringe manufacturing site until 1996, it moved into producing pen needles for insulin injection. It is now “the world’s largest pen needle manufacturing plant” (operating 11 different pen needle lines) and has approximately 350 employees.
Jabil
- Activities: Medical Devices – Manufacturing
- Employees in Dublin: 300
Jabil works with healthcare companies from around the world to design and manufacture medical products.
Previously Nypro Healthcare, the Bray site has been operational since 1980 and is now the company’s European Centre of Excellence. At this facility, they work on a range of activities such as proof of concept, prototyping, supporting clinical trials, and validation – leading to full manufacturing.
This site houses 14 clean rooms, 65 injection moulding machines and 18 assembly cells. Annual production is estimated to be 1.7 billion products and components.
In 2021, the site won the prestigious Shingo prize for Organizational Excellence.
Take a closer look at Jabil in Ireland…
Siemens Healthineers
- Activities: Medical Devices – Medical Diagnostic Equipment
- Employees in Dublin: 450
Siemens is the biggest engineering company in Europe covering sectors including energy, industry, and healthcare. The healthcare division – Siemens Healthineers – focuses on equipment for medical imaging, clinical IT, and lab diagnostics.
Siemens Healthineers is the 4th biggest Medical Device company in the world (based on 2022 revenue).
Siemens has over 600 employees across Ireland including staff in headquarters and sales roles. Siemens Healthineers has a manufacturing plant for diagnostic equipment in Swords, Dublin that is described as a “state of the art manufacturing facility producing and supporting complex medical diagnostics analysers for a worldwide market.”
In January 2023, the company announced a new “centre of excellence for immunoassay instrument research and development” that is expected to bring 100 new jobs to the site.
West
- Activities: Health Care and Medical Devices and Assemblies
- Employees in Dublin: 650
West Pharmaceutical Services design and produce “integrated containment and delivery systems for injectable medicines”. They work closely with biotechnology, medtech, and pharmaceutical companies to develop and manufacture these systems for new products.
West has had a site in Dublin since 2005. The Mulhaddart facility offers a “single-source solution” by performing design, development, and manufacturing activities. The facilities here were recently expanded to increase manufacturing capacity.
Find all of Dublin’s pharmaceutical and medical device factories on this interactive map.
(Best viewed on a Desktop or Tablet)
FAQ: Pharmaceutical and Medical Device Manufacturing Industry in Ireland
What pharma companies are in Dublin?
Dublin is home to many prominent pharmaceutical companies. Some of the key ones include:
- Pfizer – Operates a biologics manufacturing site in Grange Castle.
- Bristol Myers Squibb – Specializes in biologics manufacturing.
- AstraZeneca (Alexion) – Focuses on biologics.
- West Pharmaceutical Services – Designs containment and delivery systems for injectable medicines.
- Regeneron Pharmaceuticals – Runs business operations in Dublin.
- MSD – Has six sites across Ireland, including Dublin.
- Novartis – Manages commercial operations, marketing, and sales.
- Bayer – Focuses on marketing, sales, and regulatory affairs.
- AbbVie – Has one manufacturing site. Also oversees commercial operations and engages in R&D collaborations.
- Sanofi – Handles marketing, sales, and patient support programs.
- Eli Lilly – Manages commercial operations and supports R&D initiatives.
What are the top 10 pharmaceutical companies in Ireland?
- Pfizer: $58.5 billion (€53.6 billion)
- Manufacturing: Biologics, Vaccines
- Commercial Operations: Marketing, Sales
- Johnson & Johnson (J&J): $54.8 billion (€50.2 billion)
- Manufacturing: Medical Devices, Pharmaceuticals
- Commercial Operations: Marketing, Sales
- AbbVie: $54.3 billion (€49.8 billion)
- Manufacturing: Biologics
- Commercial Operations: Marketing, Sales
- Merck & Co. (MSD): $53.6 billion (€49.2 billion)
- Manufacturing: Vaccines, Biologics
- Commercial Operations: Marketing, Sales
- Roche: $48.7 billion (€44.7 billion)
- Commercial Operations: Marketing, Sales
- Diagnostics: Medical Devices, Lab Services
- AstraZeneca: $45.8 billion (€41.95 billion)
- Manufacturing: Biopharmaceuticals, Vaccines
- Commercial Operations: Marketing, Sales
- Novartis: $45.4 billion (€41.3 billion)
- R&D: Oncology, Immunology
- Commercial Operations: Marketing, Sales
- Bristol Myers Squibb: $45.1 billion (€41.0 billion)
- Manufacturing: Biologics
- Commercial Operations: Marketing, Sales
- Sanofi: $40.6 billion (€38.5 billion)
- Manufacturing: Vaccines, Biologics
- Commercial Operations: Marketing, Sales
- GlaxoSmithKline (GSK): $37.8 billion (€35.8 billion)
- Commercial Operations</stro
Interested in a Pharma Manufacturing Career?
No previous industry experience or science or engineering background?
Start with our Conversion Course into Pharmaceutical Manufacturing to retrain for an entry-level job.GMP Manufacturing experience? (e.g. pharmaceutical, med device, nutritional, beverage, food or dairy manufacturing)
Take our award winning NFQ Level 7 Certificate in eBioPharmaChem (15 ECTS) to move into more senior roles.Already working in the industry and want to upskill for a better job or a promotion?
View all 13 Online Pharmaceutical Courses.
You Might Also Be Interested in:
- Search Ireland’s Largest Pharmaceutical Jobs Board
- Top 10 Pharmaceutical Companies in Ireland
- See 213 Pharma and Med Device Factories in Ireland listed by county on this table
- Top Pharmaceutical Companies in Cork
- Top Medical Device & Pharmaceutical Companies in Galway
- Top Pharmaceutical Companies in Limerick
- Top Pharmaceutical Companies in Waterford
If you know of a company or group we’ve missed from this page, or think your company should be displayed here, please contact us.
About the Author
Claire Wilson
Content Marketing and Career Coaching
Claire runs GetReskilled’s Advanced Career Coaching Programme – our specially devised job hunting course that helps our trainees take that final step into employment by leading them through the job hunting process. She is extremely enthusiastic about helping people reach their final goal of employment in their new career path.
Claire has a BSc (Hons) in Medical Biology from Edinburgh University and spent 7 years working in the pharmaceutical and medical device industries.


Post Your Comments Below