You should do more than just plan for your first job in the pharma industry – you should plan for a career.
Many people make the mistake of stumbling through their career – waiting for opportunities to present themselves and for others to notice their talent.
Don’t make that mistake. Instead, aim to progress through your career by making deliberate choices to reach your goals.
Click on the links to jump the engineering discipline you’re interested in.
What is Career Planning?
As you start out in this career, you should take some time to consider where you want to end up, and plot a path to get there. It doesn’t mean that those choices are set in stone, you can still be flexible if you discover other opportunities later. But it means you have a clear goal to work towards.
And this isn’t just a task for aspiring CEOs – whatever your ultimate career goal, you should be actively working towards it.
But that’s a big task if you’ve never spent time doing something like this before. So grab a piece of paper and a pen, or open a new word document, and let us guide you through the career planning process in 5 steps…
Step 1 – Identify your motivations
In this first step, we’re going to dig deep into what you want.
Essentially you’re going to consider what kind of life you want.
This has to be your first consideration because it will change all future decisions.
So first, think about what you value most in life.
What you want from life…
What do you need from your life to feel happy?
And note that this is a deliberately bigger question than “what do you want from your career” – take a step back from that just now and think about the biggest picture.
There are no wrong answers and you don’t have to share your thoughts with anyone. So take your time and be honest (lying to yourself here is just going to take you down the wrong path).
To give you some ideas, you might consider your thoughts on:
- Money
- Travel
- Power / control
- Responsibility
- Staying within one geographic area
- Time with your family / friends
- A hobby you love
This is by no means an exhaustive list – your most important values might be something completely different and that’s absolutely fine (no wrong answers!)
It’s not necessarily something that you can write down in 5 minutes. Make a note of your first ideas, but then give yourself a chance to reflect on them and think about them a little more to really get to what you want.
For example…
You might initially write money.
But if you give yourself time to think about it – you might actually be looking to buy your dream house, or save for your children’s future, or retire early.
There can be important differences in those – so try to really get to the heart of what you want.
What you want from your career…
Now we need to think about career-specific things to uncover the right path. Don’t be constrained by thinking about the next job you’re aiming for – or any specific job for that matter. This is about the long-term, what you’d ideally want to be doing. So instead, think about having a magic wand, what would you want from a job?
Again, this can be difficult to do by yourself, so work through the following questions.
Let’s think about how you want your working day to look. This isn’t about specific tasks – it’s more about general activities. Use the list below to start your thoughts, choosing as many as you want (and feeling free to add multiple others), but then try to prioritise your list – so you’ve also got a sense of what’s most important.

Now answer these questions to help you identify what’s important to you in a job:
- What parts of previous jobs have you enjoyed?
- What tasks do you look forward to doing?
- When are you happiest? And can you capture that in a workplace environment?
- What hobbies do you have? And what do they tell you about your personality? (i.e. playing football – teamwork, leadership, physical activity)
- What types of tasks do you enjoy? (e.g. repetitive, predictable, projects, new/novel, etc)
- What tasks or roles do you see colleagues doing that you think you’d enjoy?
And now try to answer these questions to help you identify what you should avoid, if you can:
- What parts of previous jobs have you disliked?
- What tasks do you put off doing?
- What tasks or roles do you see colleagues doing that you’re glad you don’t have to?
Again, these are tasks that might immediately bring some things to mind. But to complete them thoroughly, you’ll probably need to give yourself time to think about them.
And finally, for this step, look at your answers and see if you can spot any patterns. Are there common themes? Or ideas that keep repeating? This will help you identify the core things that are important to you.
If you feel comfortable sharing your answers with someone else, see if they notice any patterns that you didn’t.
Step 2 – Figure out your skills
Now you need to figure out (and write down!) the skills you have.
To fully capture your current skill set, think about:
- Professional experience
- Experiences in education
- Hobbies & interests
- Social situations
You’ll probably find it quite easy to capture things like technical skills and things you have training in, and that’s important. But I can guarantee you have more skills than that.
I want you to delve deeper, and uncover the range of transferable skills you have.
Transferable skills are industry-independent – you might have heard of them referred to as “soft” or core skills.
Examples of transferable skills include:
- Written communication
- Verbal communication
- Attention to detail
- Leadership
- Problem solving
- Team working
- Self-motivation
- Organisation
- Result orientated
- Initiative
- Project management
- Supervision
To identify your transferable skills, think of the duties and responsibilities you’ve had in previous roles, and then think about the transferable skill involved.
For example…
You worked in an office and trained new colleagues when they joined. To help them, you wrote a check sheet for the essential parts of their job, and numbers of who to call if they had questions.
This displayed:
- Team working – helping new staff settle in and be successful
- Initiative – to create the document
- Organisation – to gather together all the resources they might need
- Written communication – describing things accurately on the check sheet
- Supervision – mentoring of junior staff
If you consider all previous experience in this way, you’ll quickly develop a long list of skills.
Once you’ve done that, look for patterns and repeating themes.
Which of these skills are related to the tasks that you particularly enjoy? Are there some transferable skills that don’t appear on your list at all?
And it’s also worth thinking about your particular strengths and weaknesses.
To identify strengths, consider:
- What am I praised for?
- What have managers highlighted as strengths in appraisals?
- What do colleagues come to me for?
- What subjects did you enjoy in school? (it may be a while ago, but it might help you to think about some of your core “likes”)
- Are there times you shine when others around you are having difficulty? (e.g. tight deadlines, tasks that need organised, when there’s conflict that needs resolved, de escalating complaints, etc)
To identify weaknesses, think about:
- What do I need to ask colleagues for help with?
- What have managers highlighted as areas for development in appraisals?
- What situations do I avoid? (e.g. big team gatherings, conflict, presenting, etc)
When writing these down, you don’t have to be concise – use as many words as you need to at the start to simply capture your awareness. Then you can worry about being able to concisely define it into skills and qualities.
You might find that in trying to put it into words, your understanding of it changes – that’s the whole point!
Step 3 – Get to know the opportunities / job roles
Ok, so now we step away from you, and look to the job opportunities in pharma.
I’d argue that this can be the most difficult stage. For now – you don’t know the things you don’t know. But don’t let that put you off, it’s worth giving this your best effort. And again, we’re here to guide you…
To do this, we’re going to think of industry opportunities in a funnel – we’re going to put all jobs in the top, and then narrow it down in a series of steps.

Understand industries
So let’s start broad and see if we can narrow down the specific industry at all. You’ve got “pharma” in mind, but there are still a range of similar options open to you:
- Pharmaceutical manufacturing – this is the traditional making of medicines that involves combining chemicals to make small, active molecules from scratch and packaging them in a way the body can use (like in a tablet).
- Biopharmaceutical manufacturing – this is a much newer industry that takes things found naturally in biology (like proteins and cells) and packages them into a usable treatment. While many of the same steps apply (and the regulations certainly do), some of the jobs can be a little different due to the different processes involved.
- Medical Device manufacturing – this is a diverse term that covers the manufacture of any product that diagnoses, prevents, or treats a health condition without any chemical change in the body. Think of things like heart monitors, contact lenses, or artificial joints. This is a very different industry – often following the procedures for discrete manufacturing – and therefore offers a very different range of jobs. Salaries can be lower than in pharma, but it can be easier to get an entry level position.
- Combination Product manufacturing – this industry manufactures products that combine a medical device with a pharmaceutical or biopharmaceutical product. Think of things like insulin injector pens, drug eluting stents, and metered dose inhalers.
This article has more information about the steps involved.
Depending on the type of job you’re interested in, you might not narrow down your search too much here.
Get to know departments
Now you understand what’s being made, it’s important to understand the different departments involved.
As a quick overview, departments include:
Research – during this stage, experiments are done to find new products or improve existing ones. This looks very different in the different industries. In pharma/biopharma, there’s a strong focus on lab-based science roles (filled by candidates with specific experience and advanced degrees). And in medical devices, engineering (including design engineering) is dominant.
Clinical Trials – all products undergo strict testing procedures to prove they perform as expected. In pharma, this involves teams of nurses, pharmacists, coordinators, and administrators.
Development – at this point testing and planning is carried out to establish how products can be made reliably at scale. There are many opportunities for engineers and some scientists at this stage.
Production / Manufacturing – this is the part of the process where the products that eventually end up with patients are made. In a typical manufacturing plant, this department makes up about 50% of staff. There is a wide range of positions available to suit many skill sets – from entry-level operator roles, through to specialist engineers and management. There is significant scope for progression within this department but it can also serve as a starting point to gain “on the floor” experience, before moving into more advanced roles in more specialist departments.
Validation – this is a specialist department that assesses and documents all parts of the manufacturing process to ensure that products are made consistently and reliably. Work in this department typically requires staff to have a detailed understanding of the manufacturing process as well as the requirements of validation (you can read more about that here). Depending on the size of the company, and the scale of the validation project, validation professionals might be employed directly by a company, or hired in as a contractor through an engineering consultancy.
Quality – this is another specialist department that sits alongside production, with a couple of notable sub-specialities.
- Quality Assurance professionals take a largely preventative role. They create, revise, and implement documentation that ensure that the manufacturing process is operating as it should. Doing so requires detailed knowledge of the manufacturing process and regulatory requirements.
- Quality Control professionals sample and test the manufacturing output at several stages, including the finished result. In pharma, Quality Control testing is typically carried out by scientists in a lab setting.
Regulatory Affairs – another highly specialised department, this time dealing with the documentation that is required by regulators. Entry-level roles are not common, and professionals typically end up here after significant experience in other departments.
Operations – this department ensures the functioning of the manufacturing plant and its equipment. There are a range of roles available here from entry-level technical roles, through to specialist engineering opportunities and management.
Sales & Marketing – often based within company head offices and sometimes as a customer-facing sales team, this department advertises and promotes the products in line with all applicable rules and regulations.
Check out this article for some additional information and example job titles for each department.
Consider your own area of interest
With your new-found understanding of the industries and departments, it’s now time to think about your preferred areas of interest / specialisation for your career.
- Do you want to be researching new products?
- Do you want to follow specific protocols and manufacture products?
- Do you want to be involved in checking product quality?
etc
Generally, the more specialised the department, the higher the salary BUT the more time, effort, and education needed to get there.
You might also spend some time researching if there are any specific skills shortages in your country (or geographic area). National level skills reports can be good for a big-picture understanding, but monitoring local jobs boards and talking to local recruitment consultants can be useful to understand the on-the-ground situation.
Step 4 – Plot a career path
Research specific roles
The easiest way to do this is to look at job adverts within your area of interest.
For example, say you want to work in quality control.
You’d start looking at as many job adverts as you can for quality control jobs of all levels. Before long you probably have an idea of the most junior quality roles, and what the entrance requirements are. And you can research up through seniority to see where your “dream job” lies.
For example, perhaps you want to maintain a “hands on” involvement with testing, you might find out that team leader level roles are where you want to end up, because any more senior and your focus would be entirely on personnel management and strategy.
While the point here is to consider any and all opportunities, this is also the time to start narrowing down your ideas based on your skillset, values, and the level of education you’re willing to pursue.
For example, if many research roles in pharma require a scientific PhD, and you don’t even have a scientific Bachelors, you’re 7-10 years away from even getting started.
Having said that, if you really like the sound of production management roles but they require a Bachelors degree and you have high school level qualifications, this is the sort of educational requirement that could be overcome with part-time study over several years while you gain work experience.
You’ll also start to notice here that different companies can call the same job different things, and that the same job title can mean different things in different companies. It can be frustrating but the only way to get past this is to read job adverts.
No one’s going to sit you down and tell you all this, you have to be willing to put in some effort.
Plot the path
With an idea of your area of interest and some sense of the level you’re ultimately aiming for, you can now start to plot a career path.
- At one end of the document, write down your dream job – it’s best if you can find a few job adverts to give you an accurate idea of an average, rather than just one company’s idea.
- Note down any essential academic/professional criteria
- Note any desired academic/professional criteria
- Write down any transferable skills mentioned as important
- At the other end of the document, write down the job that you think is your most likely point of entry to the industry. Remember that your likely point of entry may be in a totally different department from where you want to end up. For example, perhaps you want to end up as a validation lead but you currently have no relevant qualifications and no industry experience. Your likely point of entry might be as a manufacturing operator since you have the qualifications needed for that and there are opportunities that don’t require previous industry experience. Again:
- Note down any essential academic/professional criteria
- Note any desired academic/professional criteria
- Write down any transferable skills mentioned as important.
- Plot a path of jobs that connect the two.
This might be as easy a tracking one vertical path through a department with increasing seniority. But it might also involve side-steps into other departments to gain necessary experience, skills, or knowledge.
This flow chart from SkillsFuture in Singapore gives some idea of the sideways and upward movement between job roles (just note that they separate their departments out a little differently to us). You can zoom in on this image to see some of the smaller details, or head to their website to download a copy for yourself.
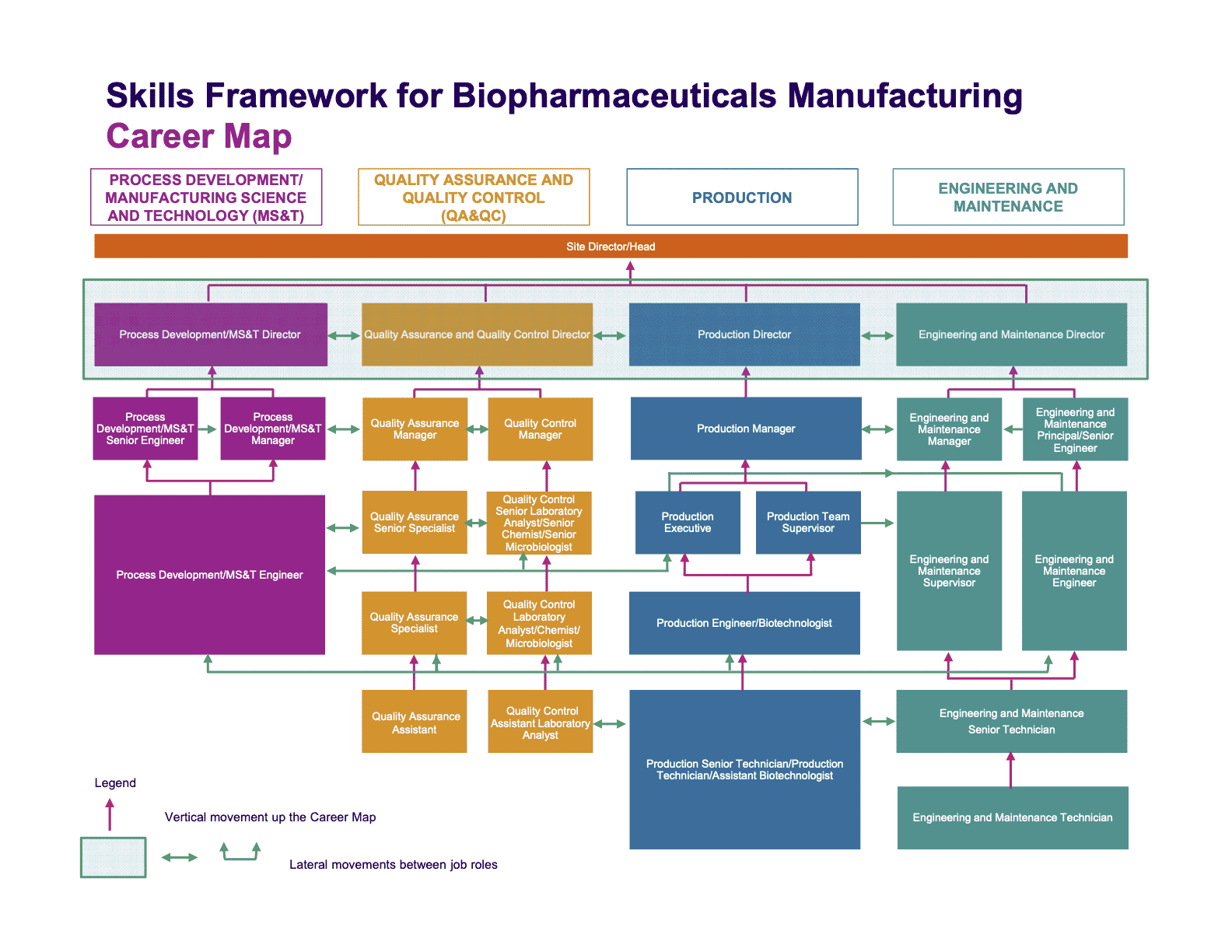
For each job you add to your path:
-
- Note down any essential academic/professional criteria
- Note any desired academic/professional criteria
- Write down any transferable skills mentioned as important.
You should end up with something that looks something like this:
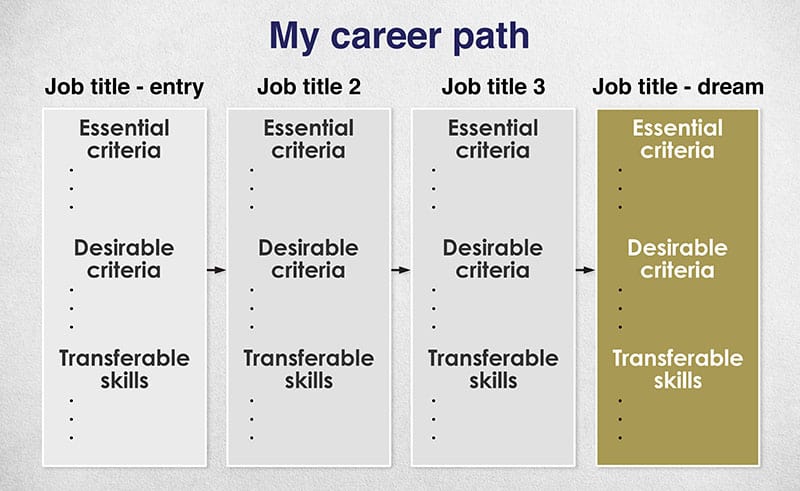
The purpose of this is to give you a realistic sense of:
- The path
- How long it will take
- What qualifications you need along the way
- What skills you need to develop along the way
Step 5 – Plan for the next job on the path
With a clear idea of where you’re ultimately headed, it’s now time to focus on the next job in the path.
You need to figure out your currently suitability to fulfil the criteria and skills you noted, and what stands between you and that next job.
Is it just time and experience? Or do you need a qualification, additional skills, or specific contacts?
To figure out, conduct a SWOT analysis on yourself.
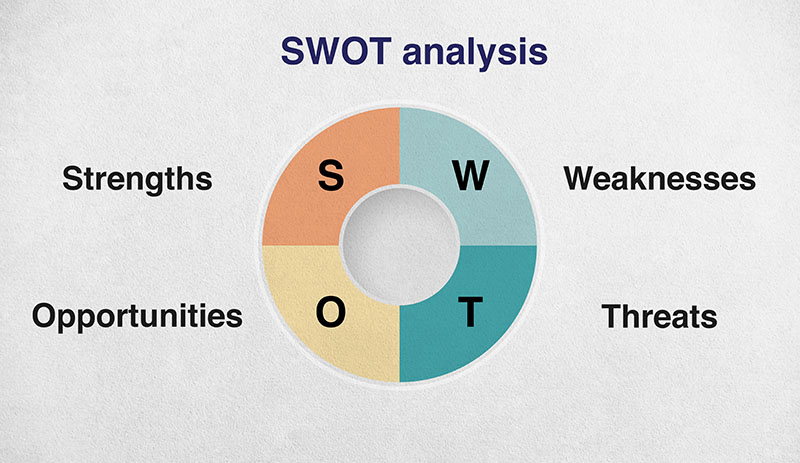
Try where possible to be objective about this, and consider things from an employers point of view.
Strengths
(be honest – this isn’t the time to feel self-conscious and no one has to see what you write)
- What are the specific useful skills and qualities you’re bringing to the next role?
- Which essential or desired criteria do you already meet?
- Why are you better qualified than your peers who might also be looking for that next step?
- What are the specific examples of success or achievement you can give that demonstrate your suitability to perform the duties?
Weaknesses
(again, be honest – this isn’t the time to try to blag your way through – it’s important that you’re aware so you can take steps to work on these weaknesses)
- What are you currently lacking that would stop you from getting that job if you applied today?
- Are you missing qualifications?
- Have you been told there’s a skill you need to develop?
- Is there on-the-job experience and knowledge you need first?
- Do you need better relationships with colleagues, management, or customers?
Opportunities
- Is the department or company expanding?
- Are there other companies nearby?
- Have any new companies been announced within your area?
- Is there an identified shortage of this job role?
- Have you made connections with people who will let you know about opportunities as they come up?
Threats
- Is the department, company, or industry slowing down in your area?
- Are there changes in the job that might leave you less qualified (e.g. changing essential qualifications, changing technology, etc)?
- Are you limited by geography?
- Do you anticipate any life events that might impact your progress towards this next role?
The next job on your path might be a few years away, so set yourself shorter term goals to make sure you’re keeping on track. Think about:
Qualifications – talk to your manager about training options. Take advantage of in-house training and keep an eye on other training opportunities (your workplace might have funding for external courses). But it might also be worth funding training or education yourself if it’s going to help you reach the next step and your workplace is resistant to fund it.
Skills – look for opportunities within and out with work. Consider building transferable skills in other places, even if your options at work at limited.
Networking – build your network as you go. LinkedIn is a great place to do this online but take advantage of in-person networking opportunities as well. Network with people on your path, recruiters, managers, etc. But be genuine, add value where you can, and be gracious to those looking to network with you because you’re further ahead on the path they’re pursuing.
Knowledge – follow news items, industry publications, groups, social networks etc where you can read about issues important to your career path. Don’t wait for someone to tell you, take initiative.
Set SMART goals
If you need a minimum 3 years experience in this job to be considered for the next, how will you know you’re on track? What do you want to have achieved by next year to know that you’re still on the right path?
Set some annual goals, but think about how this maps across the year. You need to keep making progress. Set smaller goals so you keep the feeling of momentum and progress.
Let’s take the goal of “I’m going to be a better employee”. That’s not a very good goal. What does “better” look like, and when will you know when you’re achieved it?
Goals should be SMART…
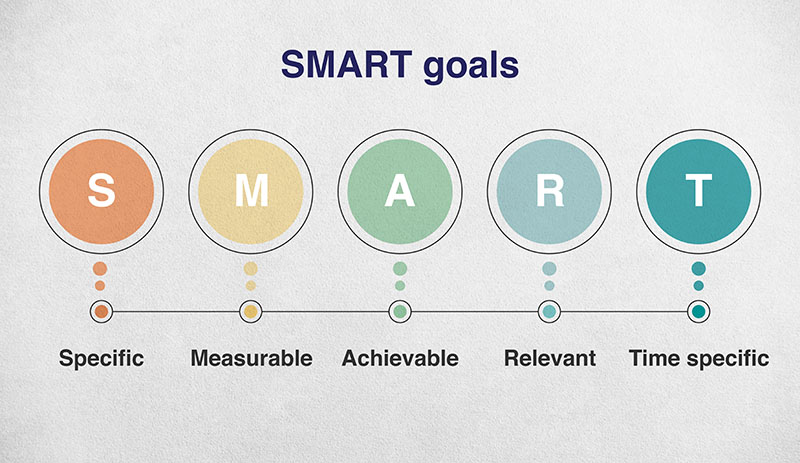
So let’s improve that goal by making it SMART:
Specific – goals can’t be broad, you have to clearly know when you’ve achieved them.
E.g. move from “I’m going to be a better employee” to “I’m going to be a better employee by being off less, getting on better with my colleagues, and increasing my job knowledge”
We’re being specific about what a “better” employee looks like.
Measurable – goals should involve a specific metric that you can track and see the progress in.
E.g. move from “I’m going to be a better employee by being off less” to “I’m going to decrease my absentee rate to 0”
We’re introducing a metric we can measure, and a target to aim for.
Achievable – there’s no point in setting goals you can’t achieve, you’ll just get demoralised, and perhaps even stop trying to make progress at all.
E.g. move from “I’m going to decrease my absentee rate to 0” to “I’m going to decrease my absentee rate by 90%”
It’s still specific and measurable, but it’s not going to feel completely lost if you get the flu and have to take some time off.
Relevant – your goal should have a direct impact on the thing you’re trying to accomplish. There’s no point in putting lots of effort into something, if it’s not ultimately going to do you any good.
E.g. we could set a goal of “bringing lunches from home every day”, and while that might achieve a lot of things, it’s unlikely to be relevant to getting the next promotion
Time specific – you have to create a timeline, so you’ll know when you’re aiming for and you’ll know if you’ve made it.
E.g. move from “I’m going to decrease my absentee rate by 90%” to “I’m going to decrease my absentee rate by 90% this calendar year”
Start keeping proof
Competency based interviewing is routinely used throughout the pharma and med device industries. The typical “Tell me time a when…” questions can be challenging if you’re not prepared.
But with the research you’ve done to get to this point, there’s no excuse.
Competency based questions usually assess transferable skills and since you’ve already identified the common skills required for your next job, you’re one step ahead.
Long before you’re sat in that stressful interview chair, take notes of examples of key competencies, keep track of successes, and record details of your achievements.
Keeping a file of emails that praise your work, training certificates, and copies of appraisals is also a great idea.
Doing this now means that any future interview preparation should be much easier, and you’ll be ready to jump on an opportunity at short-notice, if you need to.
Step 6 – Keep your plan live
Revisit it
Revisit your career plan regularly. Schedule it, add it to your calendar, set a reminder for it.
Find a check-in timeline that works for you. Some people like to revisit goals weekly to make sure they’re moving forward and there’s no sense of “wasted time”. For others, that’s too much, and monthly check ins are preferred.
Appraisal preparation can be a good reason to revisit it, but you probably want to check in on your progress towards your goals more frequently than annually. What’s most important is that you pick a timeline that you’ll stick to.
When it’s time to review your career plan, revisit it with updated knowledge and information to consider:
- Are your goals still the same?
- Is the route to get there still the same?
- Have you found a better option?
- Or a quicker path?
Once you’re still sure you’re headed in the right direction, update your CV, LinkedIn profile, and collection of examples.
Be on the lookout for opportunities
Always be keeping a lookout for opportunities to make the next step. Your company might have a good development plan in place, but that puts you on their timeline. It might be quicker to move companies for promotions.
Keep some flexibility. The plan’s not about deciding on a goal job and rigidly sticking to that idea for the next 10 years, it’s about giving you a long term focus so you can keep momentum. It’s fine to change things as you get more information.
And if you do get opportunities, compare them against your goals. Does it help you reach your goal? Are you consciously changing your goal? Be deliberate in your decision making.
Talk about your plans
The final part to keeping your plan alive is to talk to others about it.
A good manager will want to hear about goals that they can help you work towards. Use appraisal time to discuss the next steps and what they can do to help get you there. Agree action plans, and make sure they’re followed up.
But more than that, talk to people more generally about your goals. People can’t help if they don’t know what you’re aiming for. This TEDx talk gives a great overview…
Final thoughts
So there you have it.
6 steps to career planning.
It might seem a little daunting, but there’s nothing big or scary in each step. Just take your time, put the work in, keep your plan up to date, and always keep striving for your career goals.
About the Author
Claire Wilson
Content Marketing and Career Coaching
Claire runs GetReskilled’s Advanced Career Coaching Programme – our specially devised job hunting course that helps our trainees take that final step into employment by leading them through the job hunting process. She is extremely enthusiastic about helping people reach their final goal of employment in their new career path.
Claire has a BSc (Hons) in Medical Biology from Edinburgh University and spent 7 years working in the pharmaceutical and medical device industries.

Post Your Comments Below