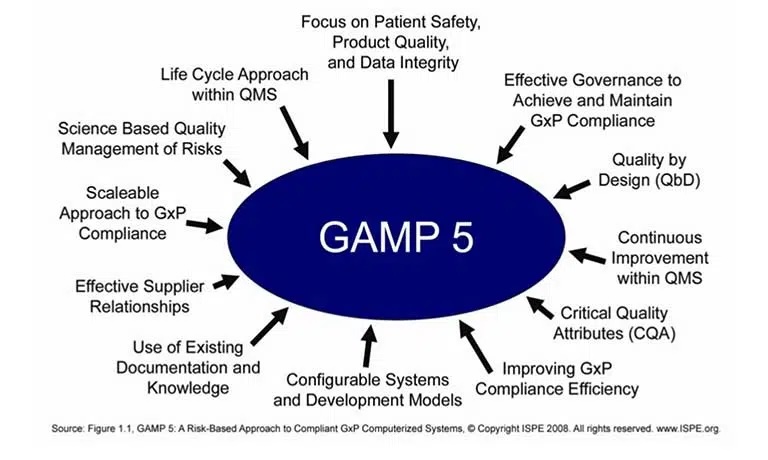
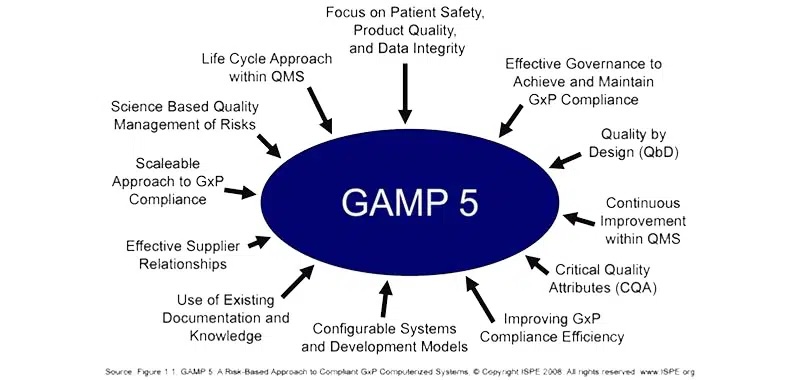
Entry Requirements
This program is highly specialized so you MUST have a solid understanding of GMP rules, regulations and guidelines.
AND you are any of the following…
- Validation Engineer/Specialist or Senior Validation Manager
- PhD, Masters or Degree in Biochemistry, Microbiology, Molecular Biology, Lab/Science, etc
- Automation, Control/Instrumentation, Process, Manufacturing, etc Engineer or Technician
- Quality Assurance or Quality Control Technician or Specialist
- Metrology, Maintenance Engineer, Technician or Specialist
- Front/Backend Developer
- Not sure if you meet the minimum requirements? Contact us.
Add On Program: If you don’t have experience working in a GMP manufacturing environment, you need to start with our 4-Week GMP Training For Beginners For The Pharmaceutical Industry.
If you want to learn GAMP®5 and 21 CFR Part 11 together, then take our 10-week online program on Computer System Validation
Your 4-Week Class Schedule
Your Class Director will check your study logs and only will release subsequent week’s materials to you if you have been logging in for more than 10 hours per week.
And he or she will proactively follow up with you to keep you on track.
What Does the Online Classroom Look Like?

We use one centralized platform where you can log into your classroom. There you can get assignments, interact with faculty and peers, reply to message boards, and more.
Hear From People Who’ve Taken This Course

Ken O’Keeffe
“Interesting and Helpful in my Current Role”
“I have found the GAMP®5 course to be both interesting and helpful in my current role. The individual sections of the course are easy to follow. The delivery of the course is really straight forward. During the course there has been times when I have been busy with work and have not been able to study. The structure of the course allows me to be flexible in my study.”

Giorgia Romeo
“I Would Highly Recommend This Course”
“I would highly recommend anyone wishing to gain an education in the GAMP®5 to do this course. For me it has been very useful studying on-line in order to do the course in my own time. Lessons are very clear and the assignment deadlines make you disciplined to meet the deadlines set.”

John Ryan
“The Course Material is Concise and Interesting”
“The professionalism and passion of the lecturers comes across in the tutorials which gives the student confidence and encouragement to keep on top of the course. The course material is concise and interesting. The timing of the release of the lessons is perfect, the student moves at a comfortable pace, one step at a time.”
Delivered by an Industry Expert
Dr. Joe Brady
Full-Time Validation Lead
Lecturer, Technological University Dublin, Ireland
Senior Associate, GetReskilled
Dr. Joe Brady is full-time practicing Validation Lead and an assistant lecturer with Technological University Dublin (TU Dublin), in the School of Chemical and Pharmaceutical Sciences. Joe is a certified trainer and highly experienced in competency-based training. He designs and prepares educational modules and full academic courses ranging from MSc, MEngSc. BSc, to Certificate level, for a range of academic institution.
He is also a supervisor for MSc/MEngSc and PhD theses. Joe has over twenty years of project experience in the pharmaceutical, biopharmaceutical and medical device industries in Ireland, Singapore, China, The Netherlands, France and the USA.
Show Off Your New Skills: Get a GAMP 5 Certificate of Completion
Once the course is over, complete a written assignment to get certified in GAMP®5 – Good Automated Manufacturing Practice
Add it to your resume, your LinkedIn profile or just get that well-earned raise you’ve been waiting for.

Price
One Person
Price For 1-Person
$1,299
One Person
Price For 1-Person
€1,199
One Person
Price For 1-Person
£999
Try Us Risk FREE for 7 Days 100% Money Back Guarantee.
Price
One Person
Price For 1-Person
€1,199
One Person
Price For 1-Person
$1,299
One Person
Price For 1-Person
£999
Try Us Risk FREE for 7 Days 100% Money Back Guarantee.
Price
One Person
Price For 1-Person
£999
One Person
Price For 1-Person
€1,199
One Person
Price For 1-Person
$1,299
Try Us Risk FREE for 7 Days 100% Money Back Guarantee.
Application Deadline: Wednesday 20th May
Program Starts: 25th May
Start Your Application
Click below to start your application for our GAMP®5 course.
Talk To Our Team
USA/World
Call Geraldine: +1 (617) 901 9268
Ireland
Call Sinead: +353 (0)21 2409016